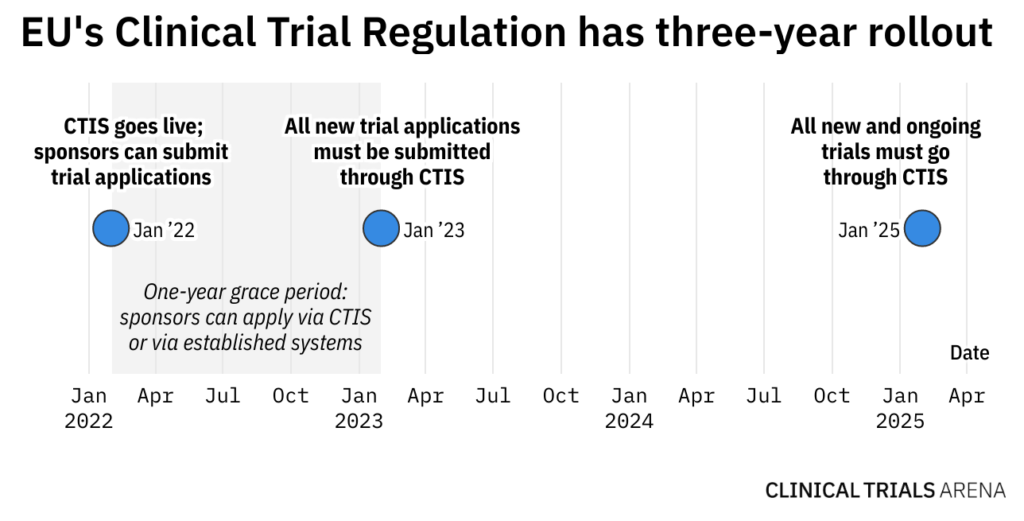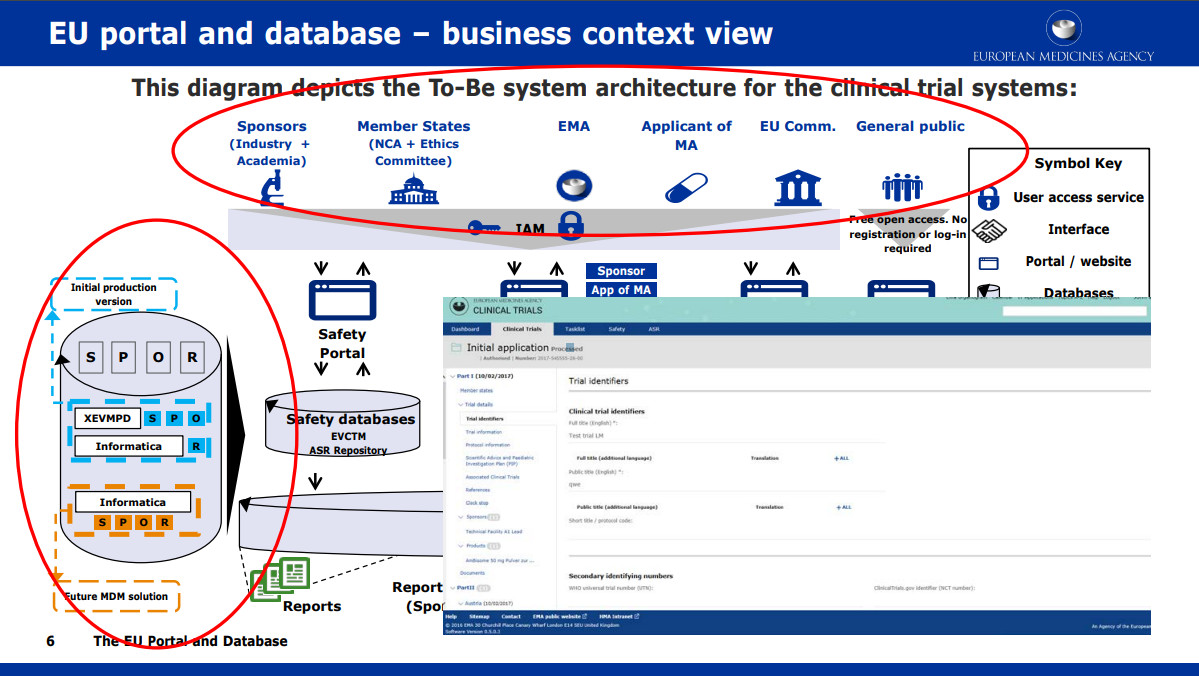
What Are the Documents Required for Clinical Trial Applications to Regulatory Authorities in Europe? - Sofpromed

EU Health - #HealthUnion a Twitteren: "Guidance on clinical trials during the #COVID19 pandemic, including key changes to: ➡️Distribution of the investigational medicinal products ➡️Monitoring ➡️Remote source data verification (SDV ...

Cochrane supports European regulators as they urge clinical trial sponsors to share their results | Cochrane

Flow diagram of the study. Abbreviation: eU-cTr, eU clinical Trials... | Download Scientific Diagram