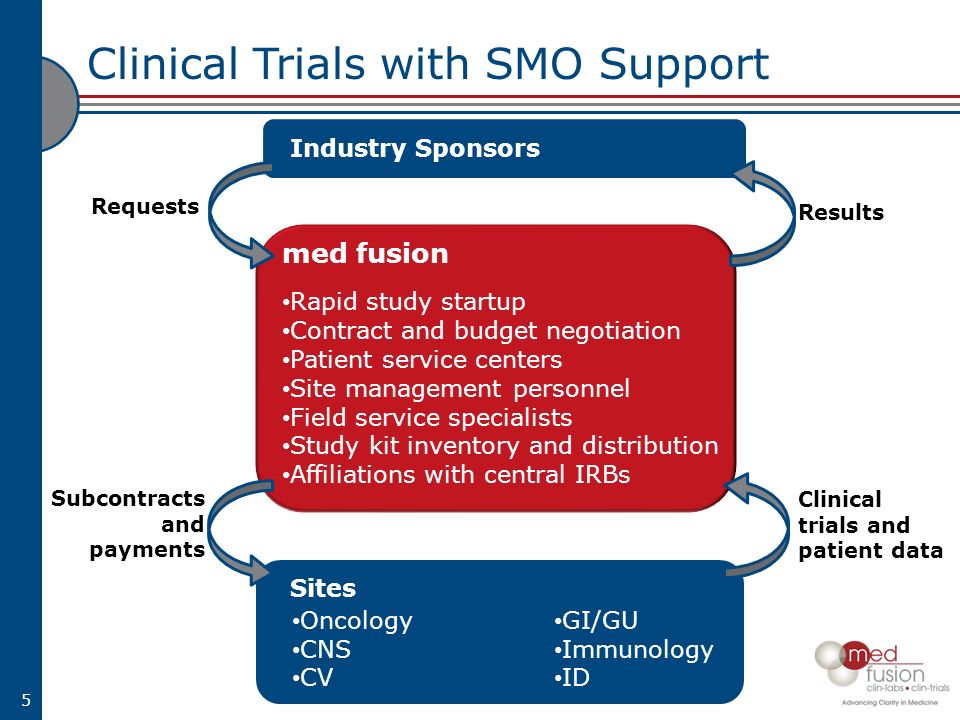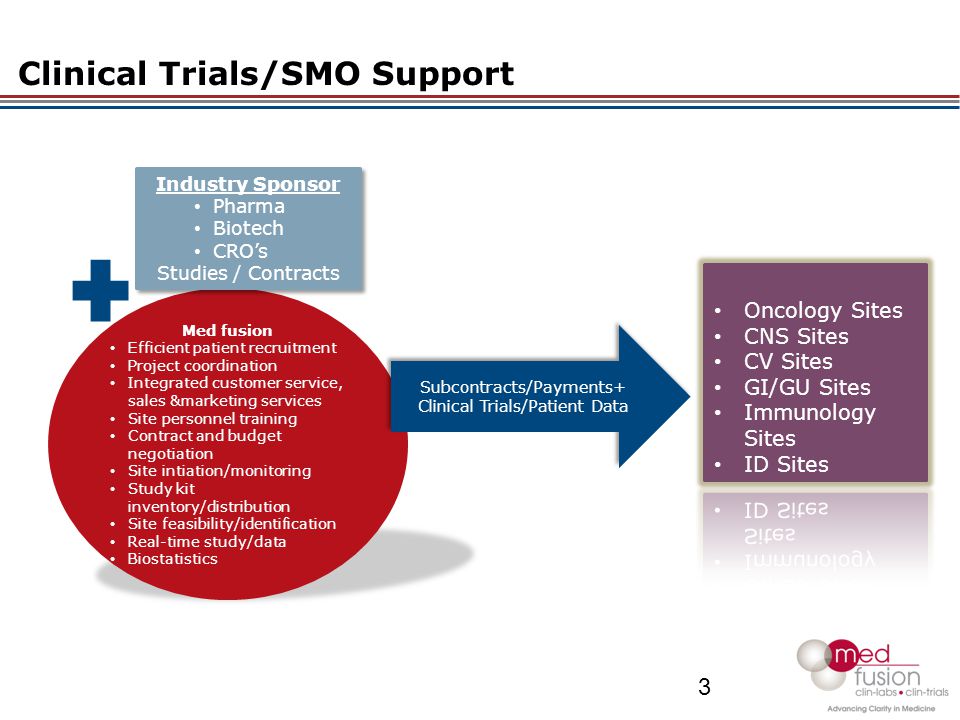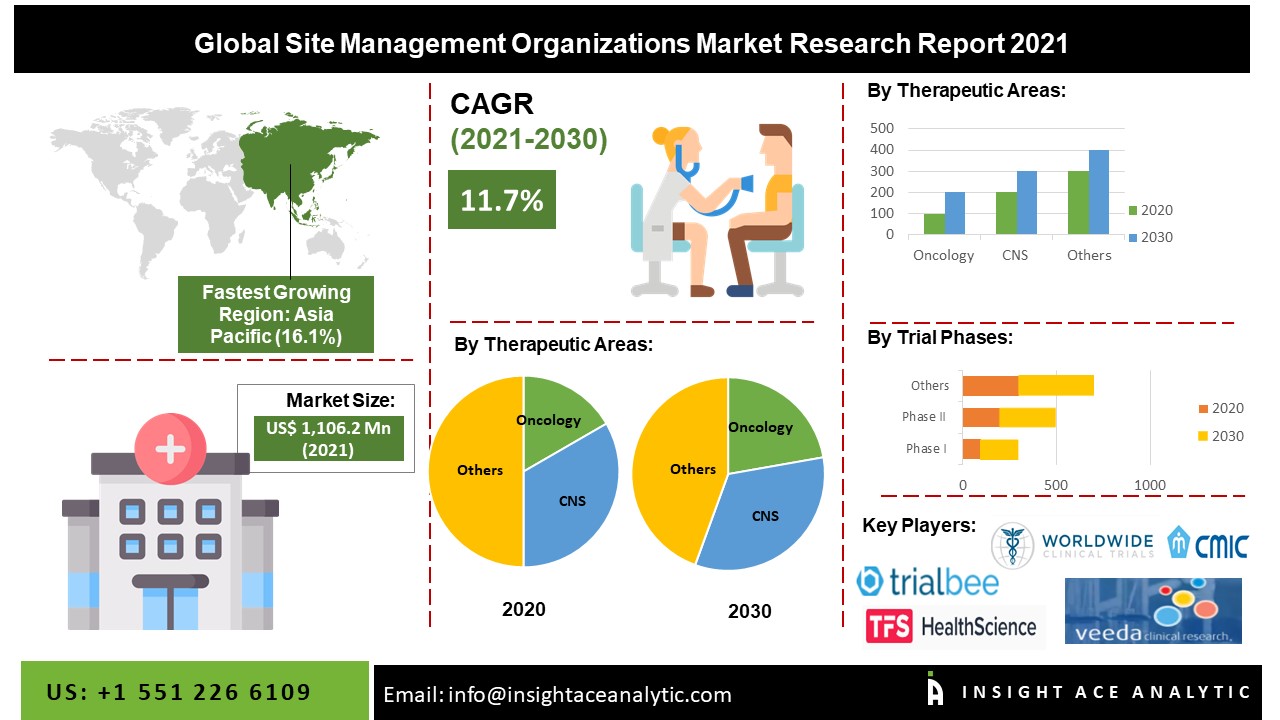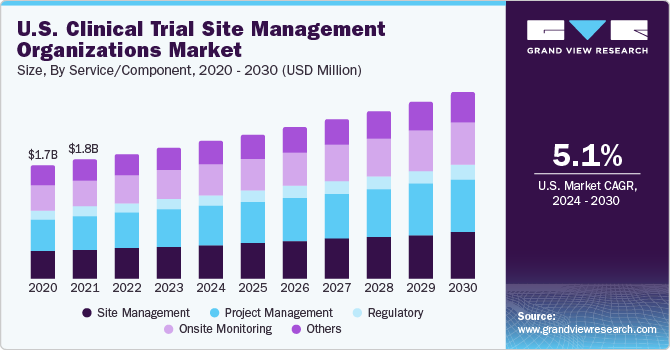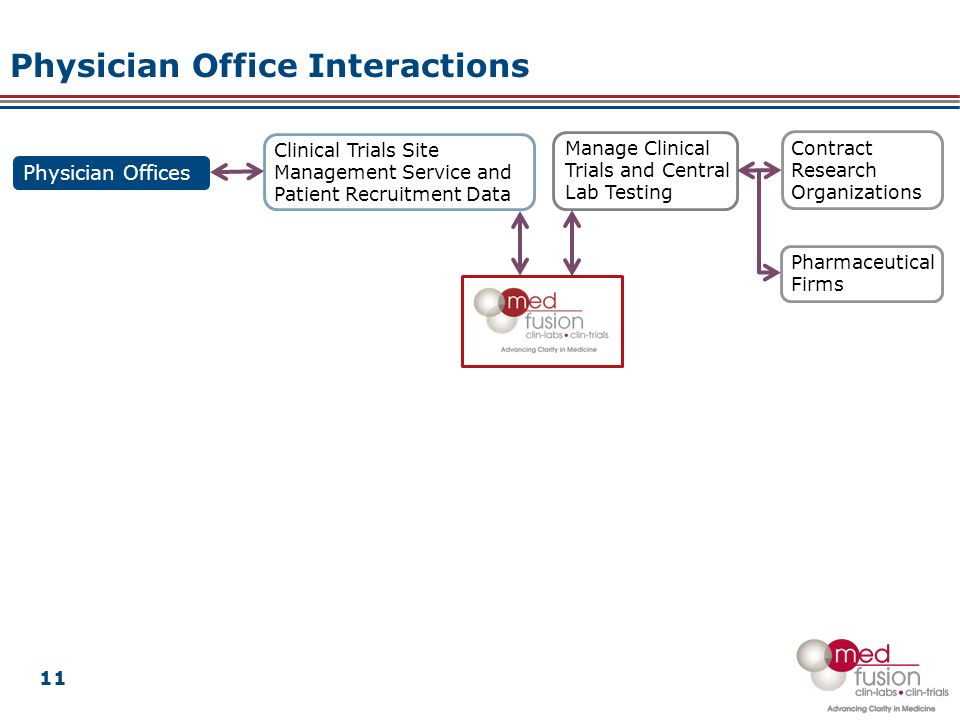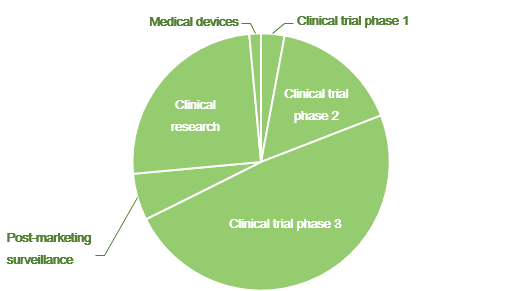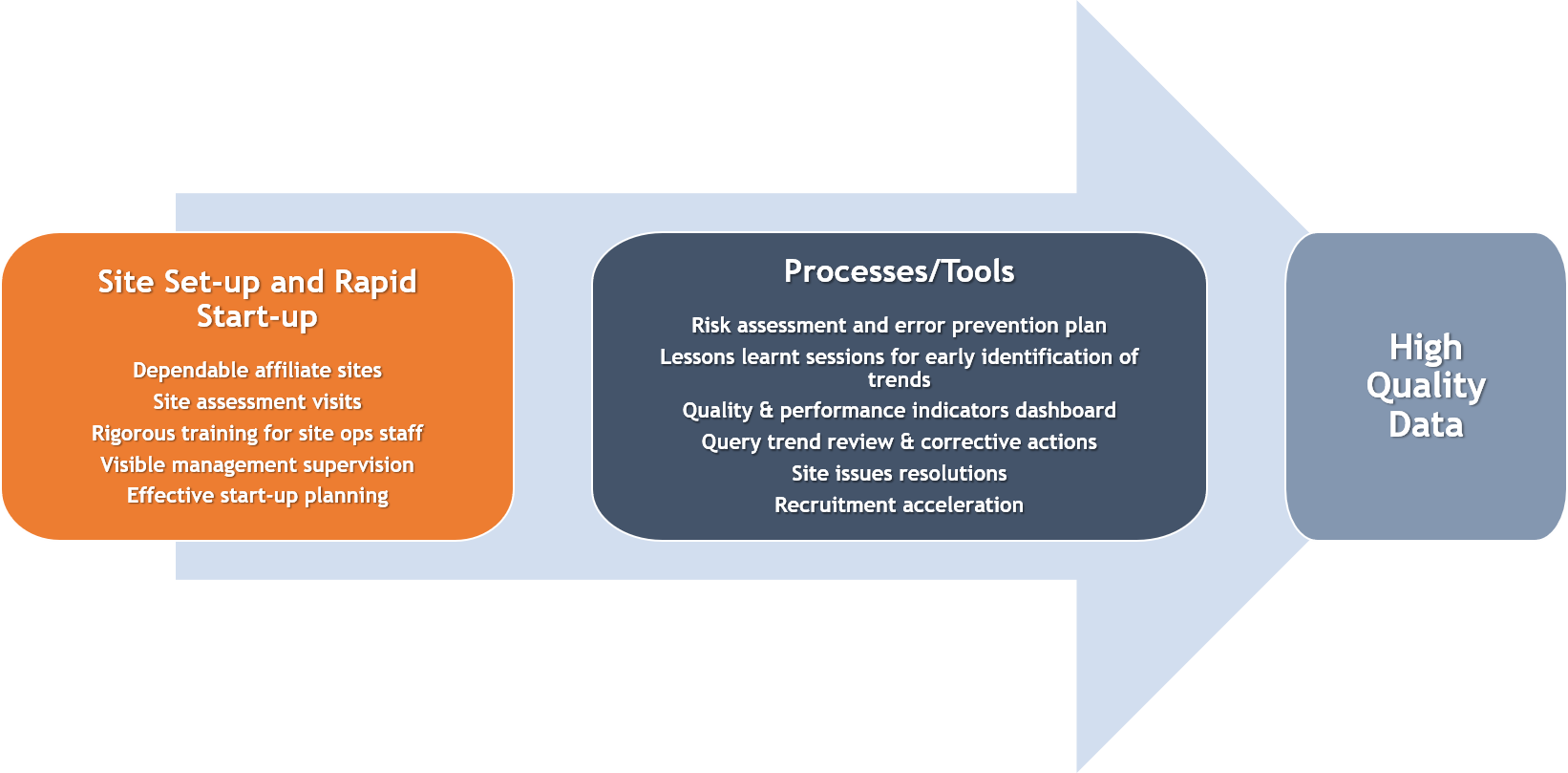
Overall Study Management | S4-Research – Site Support Services for Clinical Trials I Site Management Organization I SMO India I Site Management Organisation I SMO Asia I SMO India I SMO I

Clinerion and iHealth Group partner with Bioserv SMO to accelerate clinical research in Brazil by introducing Clinerion's PRS to the Hospital São Vicente de Paulo | World Pharma Today

Considerations for Third Party Vendor Management in a Risk-Focused Environment | Pharmaceutical Outsourcing - The Journal of Pharmaceutical & Biopharmaceutical Contract Services