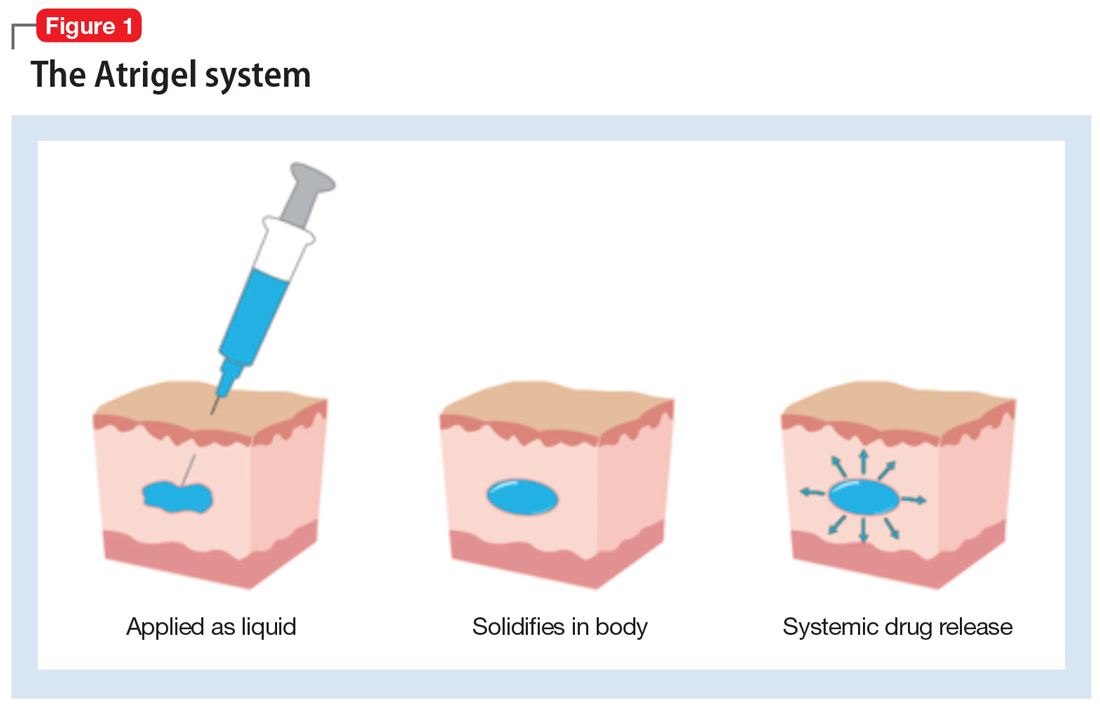
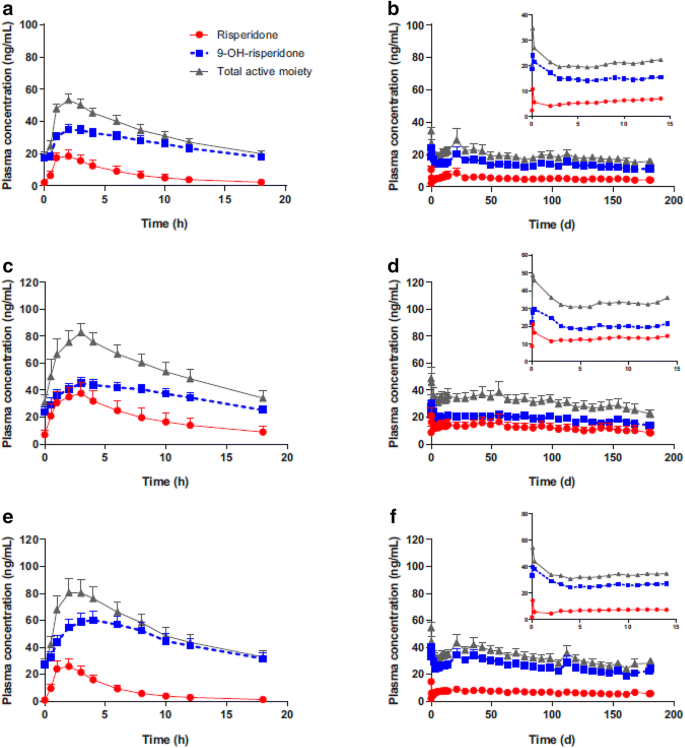
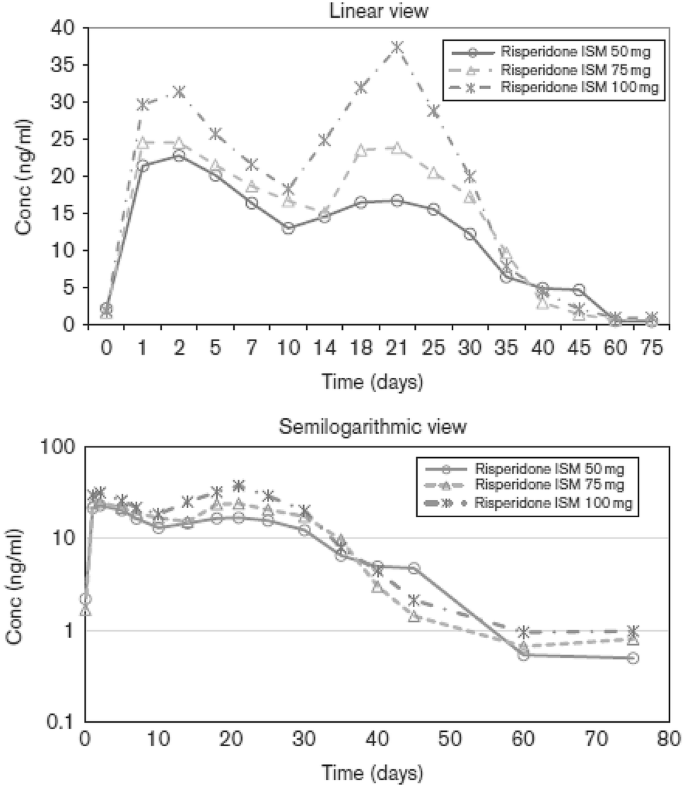
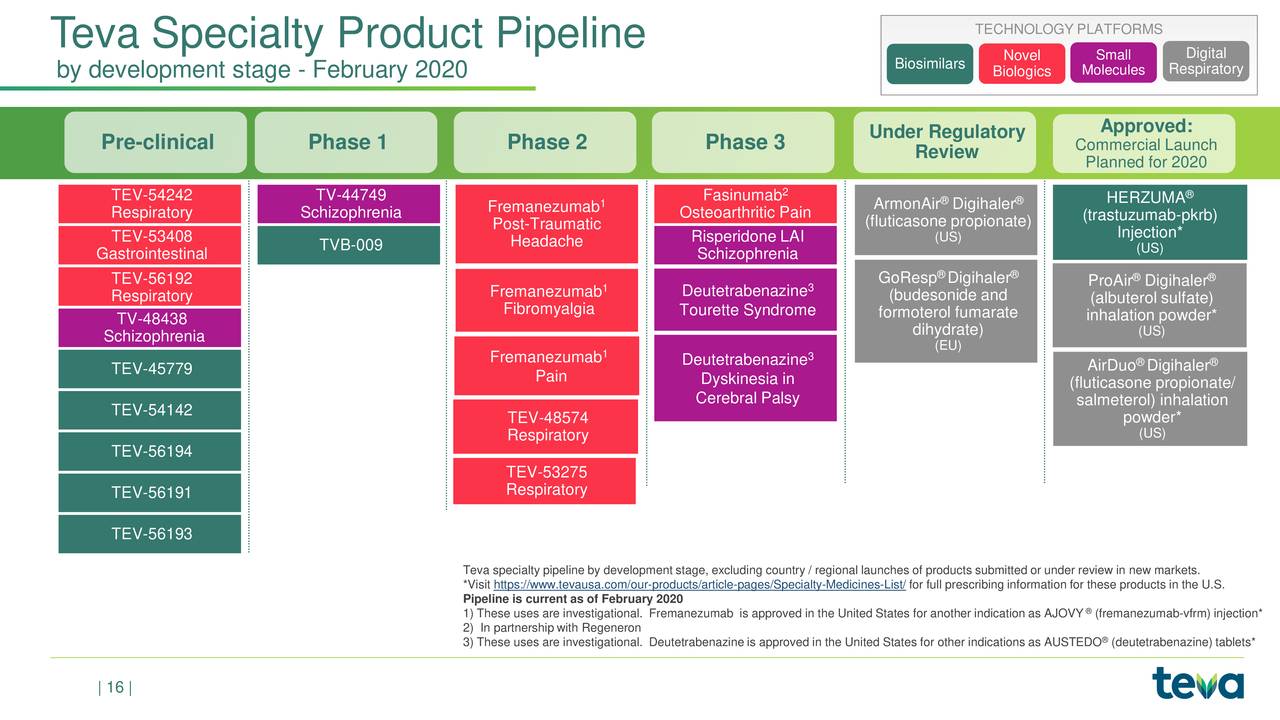
Teva and MedinCell looking to enter LAI schizophrenia market - Pharma Technology Focus | Issue 103 | February 2021

Teva, MedinCell announce positive results for registration trial of TV-46000/mdc-IRM for patients with Schizophrenia - Pharmaceutical Business review
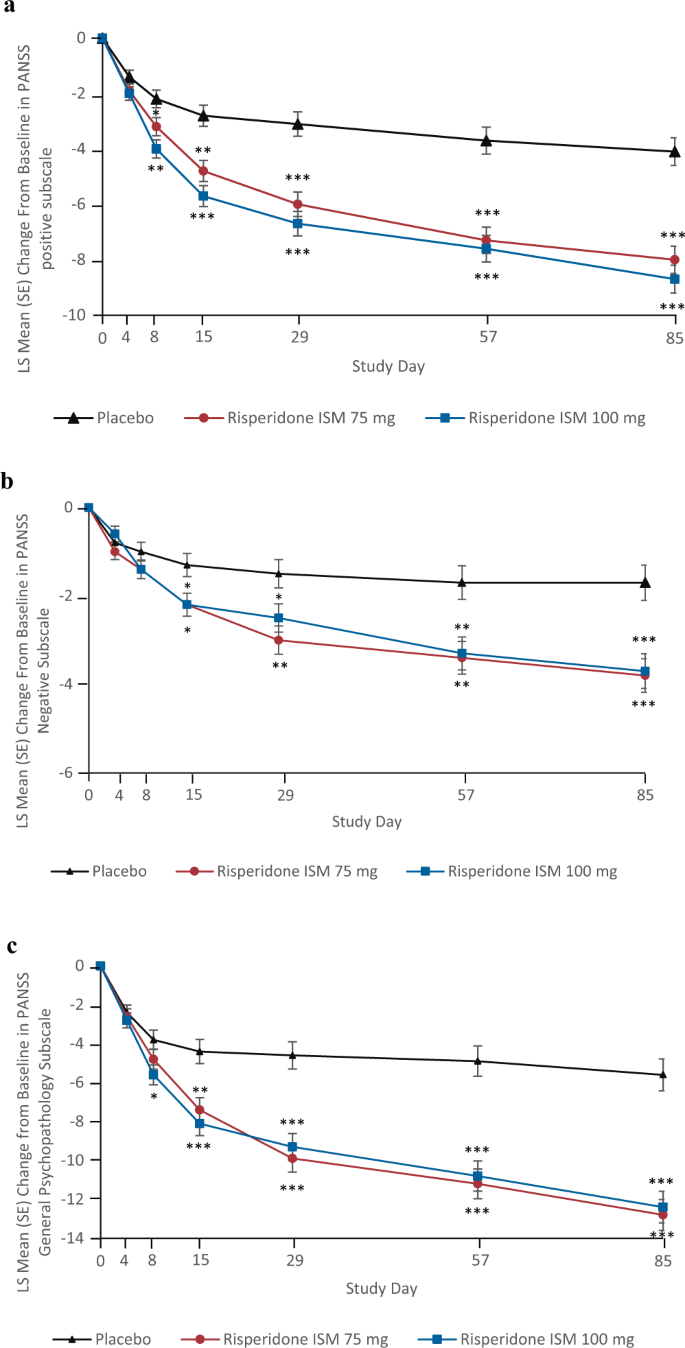
Efficacy and safety of once-monthly Risperidone ISM® in schizophrenic patients with an acute exacerbation | Schizophrenia
PROPOSAL FOR THE ADDITION OF RISPERIDONE LONG-ACTING INJECTION AND PALIPERIDONE PALMITATE 1-MONTH LONG-ACTING INJECTION FOR THE

Teva and MedinCell Announce Positive Results for Registration Trial of Investigational Extended-Release Subcutaneous Injectable Risperidone for Patients with Schizophrenia | Business Wire

Teva and MedinCell Announce Positive Results for Registration Trial of Investigational Extended-Release Subcutaneous Injectable Risperidone for Patients with Schizophrenia | Business Wire
Teva and MedinCell Looking to Enter Increasingly Competitive LAI Schizophrenia Market - Clinical Trials Arena
Study: Subcutaneous Injectable Risperidone Improves Outcomes for Certain Patients With Schizophrenia